A new weight loss pill just launched
Here's the corporate opportunity nobody sees yet
Hola amigos,
I was reflecting this morning, on the way to work, on how society lately has been celebrating rapid overnight success as if it were something to look up to.
I spent my whole life training for a Men’s Health cover I only achieved when I was 27.
I spent years counting endless reps on the gym floor, only to then become one of the first Chief Wellness Officers in global finance in my early thirties.
You know what I remember?
The hunger leading up to the shoot.
The 4am wake-ups prepping my meals for the day.
The first sip of coffee while waiting for the gym to open.
Can we please just normalize building our businesses and our lives slowly?
Can we all agree that we should not compare our day 1 with someone’s day 100?
If you are building today—whether it’s your health, your business, or yourself—remember: you are not late.
Today you will understand:
Why the approval of Foundayo, the world’s first oral GLP-1 weight-loss pill, can be a corporate wellness problem
What the rebound data actually means for workforce health costs, and why most organisations are completely unprepared
The exact move that turns this emerging drug trend into a paid corporate contract
1. The pill changes everything
On 1 April 2026, the US FDA approved Foundayo (orforglipron)—Eli Lilly’s once-daily oral GLP-1 weight-loss pill.
No injection.
No food or water restrictions.
One pill, once daily.
Within days, it was available in multiple markets.
This is the moment the GLP-1 category goes mainstream.
Until now, injectable GLP-1 drugs had a natural adoption ceiling.
Needles create friction.
Clinic logistics create friction.
Cold chain requirements create friction.
Eli Lilly’s own CEO acknowledged that fewer than 1 in 10 people who could benefit from a GLP-1 are currently taking one, held back by access, stigma, and perceived complexity.
Foundayo removes every one of those barriers.
In clinical trials, participants at the highest dose lost an average of 12.4% of their body weight over 72 weeks. The drug is already being described as a tool to “reach the planet”—Eli Lilly’s words, not mine—without the manufacturing or supply chain constraints of the injectables.
The adoption curve that has been building slowly for three years is about to accelerate sharply. And the corporate wellness infrastructure to support it does not exist.
2. The cost no one has calculated
According to Gallup data cited in the Global Wellness Institute’s Monthly Barometer from February 2026, 12% of Americans are already on GLP-1 drugs.
That figure will rise, and it will rise faster now that the pill removes the last friction point.
Here is the number that matters for corporate wellness professionals.
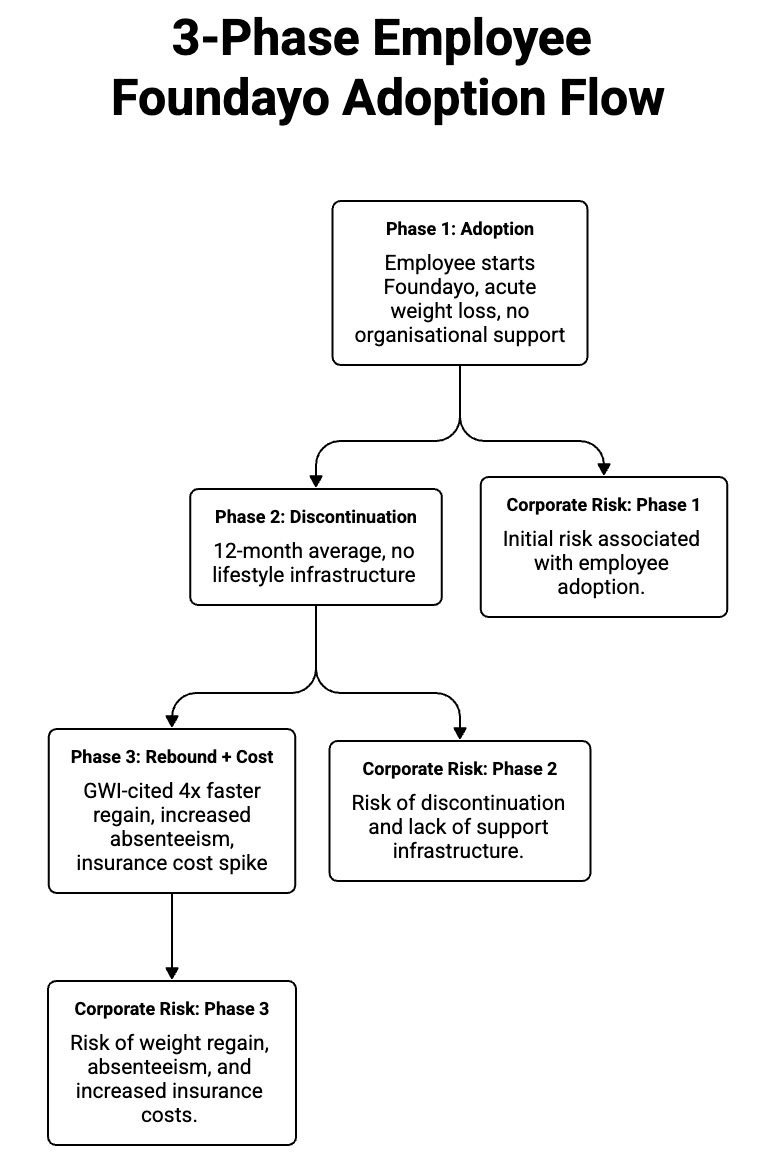
When patients discontinue GLP-1 therapy—which a significant proportion do within 12 months, due to cost, side effects, or life circumstances—the rebound effect is almost four times faster than coming off a conventional diet. This is documented pharmacological reality, not opinion.
The drug produces acute weight loss.
Discontinuation without lifestyle infrastructure produces rapid, compounded weight regain.
In some populations, the post-discontinuation metabolic markers are measurably worse than the pre-treatment baseline.
This creates a specific corporate health cost equation that almost no organization has modeled.
A portion of the workforce has started Foundayo.
A portion of those discontinue within 12 months.
Without a wraparound protocol—nutrition behavior, movement, sleep, and stress regulation—the rebound is the documented outcome.
That rebound shows up as absenteeism, insurance claims, and productivity drag 18 to 24 months after adoption began.
By the time it is visible, it is a reactive problem.
Right now, it is a preventive opportunity.
3. Wrong budget. Wrong room
Most organizations will handle this the standard way.
GLP-1 coverage is added to the insurance plan.
A memo goes out.
The wellness team is not consulted.
The medical function does not talk to HR.
That is the benefits response. It funds the drug and ignores the infrastructure condition built into the drug’s own regulatory approval.
Foundayo is approved specifically for use alongside a reduced-calorie diet and increased physical activity.
The lifestyle layer is not a wellness suggestion.
It is a clinical requirement written into the prescribing information.
Most organizations will fund the drug and ignore the condition.
The wellness professional who understands this distinction is not pitching a program. They are identifying a gap between what the drug requires to work long-term and what the organization is providing and offering to close it.
That is a risk management conversation. It draws from a different budget than a wellness program.
It goes to a different person in the building.
And right now, there is almost no competition inside it—because the drug only launched weeks ago and most wellness professionals have not connected the dots yet.
The window for walking into that room first is open.
It will not stay open.
This week’s paid brief covers the GLP-1 corporate opportunity — and it is the most time-sensitive issue I have published this year.
What follows is the internal version — not the conference version.
Three corporate interventions, ready to use.
The exact boardroom language for each one.
Three AI prompts you can deploy today.
A 15-minute briefing structure you can walk into this week.
I have done the work. You implement it.
That is the difference between reading about this opportunity and being in the room when it opens.
Less than 1 AED a day.
Less than $2 a week if you’re outside the UAE.
Keep reading with a 7-day free trial
Subscribe to Wellness Intelligence to keep reading this post and get 7 days of free access to the full post archives.